How the cilioscleral approach fits in with other glaucoma surgical techniques
- Technical Articles
Prevalence of glaucoma
Glaucoma is a group of progressive, neurodegenerative eye diseases associated with irreversible optic neuropathy followed by visual-field impairment. Epidemiological estimates suggest that the most common form, Primary Open-Angle Glaucoma (POAG), affects more than 3% of people aged 40 years or older. Prevalence increases with age. In people over 90 years of age, the prevalence is 10%. Worldwide prevalence is expected to exceed 111 million cases by 2040. According to the latest data from the Global Burden of Disease Study 2017, worldwide, glaucoma continues to be leading cause of irreversible blindness.1-5
The structural changes of the anterior segment of the eye play an important role in the subdivision of glaucoma into different types; and the anti-glaucomatous treatment is based on the underlying form of glaucoma. The physician’s first priority is the preservation of the visual function, which is achieved by monitoring the patient’s intraocular pressure. This can be done through topical medication, laser therapy and glaucoma surgery.1,6
Outflow pathways of the aqueous humor
The aqueous is formed by the ciliary body and fills the anterior chamber of the eye. From there, two pathways have been identified and widely documented in the literature:
- The ‘conventional’ outflow pathway begins in the anterior chamber and runs from here via the trabecular meshwork to the Schlemm’s canal, the collector channels and finally into the episcleral veins. These flow into the systemic venous circulation. 70% of aqueous humor outflow occurs via this ‘conventional’ outflow pathway of the eye7
- Until recently it was thought that the unconventional or uveoscleral outflow pathway collects only up to 35% of aqueous humor in the young population. For persons older than 60 years, the figure is only 3%.8,9 The uveoscleral outflow pathway was for long considered as a minor route. However, according to recent research data, the aqueous humor outflow through the uveoscleral pathway varies from 4-60% in the human eye.10 The decisive factor for the passive fluid movement is the pressure gradient that prevails here. The uveoscleral outflow of aqueous humor passes through the ciliary muscle fibers into the supraciliary space. From here it passes through emissary channels around the vortex veins through the anterior or posterior sclera or into the choroidal vessels7
A disturbed balance between the production and reabsorption of aqueous humor can result in increased intraocular pressure.12
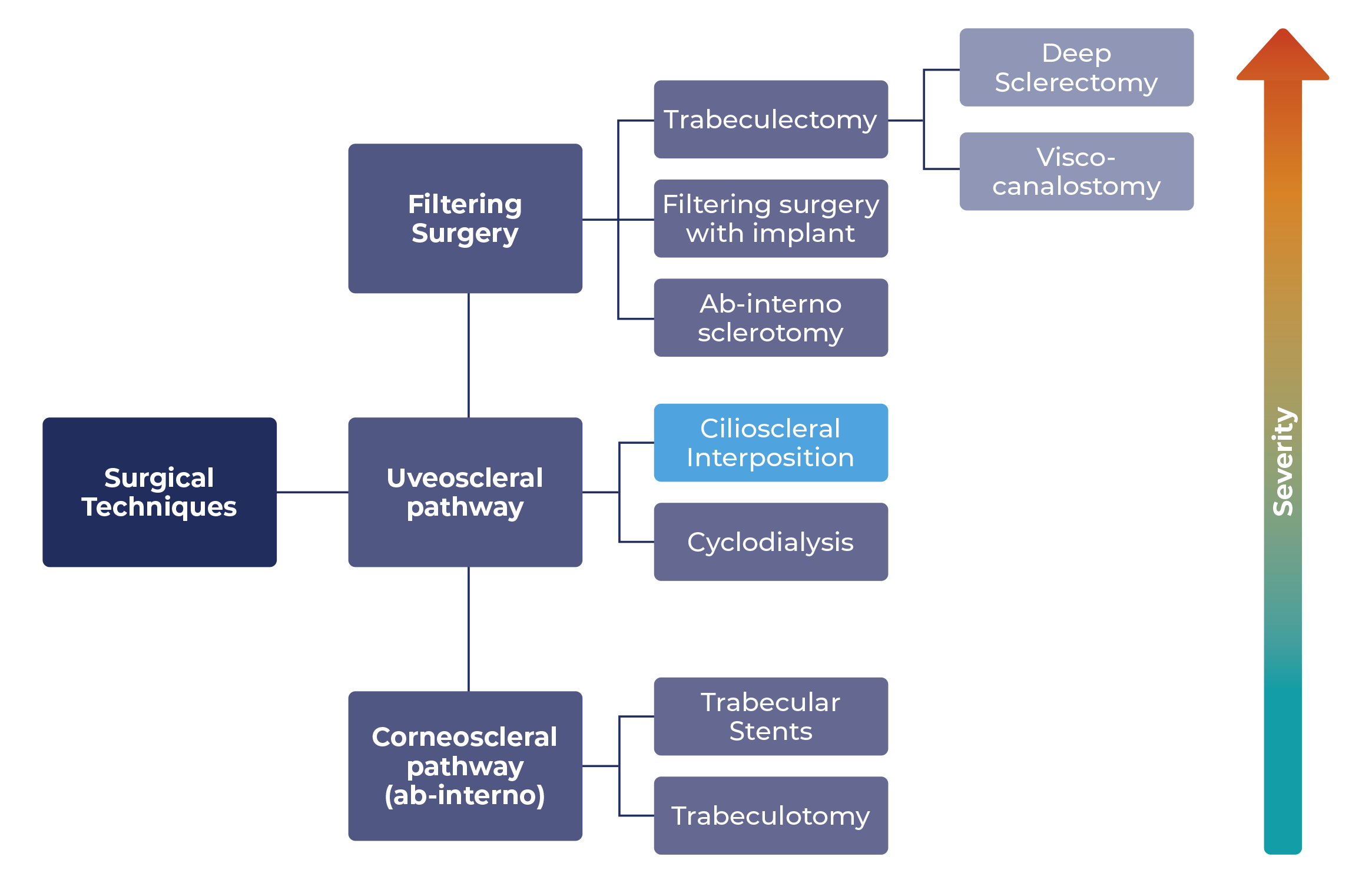
The three main surgical strategies in the treatment of glaucoma
The types of surgical techniques, including MIGS, that are applied for increasing the outflow in mild to moderate glaucoma refractory to medication and laser treatment fall into three basic categories:
a. filtering incisional surgery
b. corneoscleral pathway (ab-interno) procedures, and
c. by passing through the uveoscleral pathway
- Filtering (incisional) surgery is a group of surgical procedures which aim at creating an artificial egress from the anterior chamber through an ostium in the sclera or trabecular meshwork to a small blister of fluid created under the conjunctiva called a ‘bleb’; filtering surgeries are ‘bleb forming’. The most frequently performed filtering surgery is trabeculectomy13
- In corneo-scleral surgical methods, the anterior chamber is accessed via a clear corneal incision. In trabeculectomy ab-interno, a direct opening is made in the area of the trabecular meshwork; in this way, a direct outflow path between the anterior chamber, the outer wall of the schlemm´s canal, the collector channels, the intrascleral and episcleral vessels is formed.14,15 Trabecular stents are supposed to penetrate into the schlemm canal through the trabeculum to bypass the trabecular meshwork resistance to outflow16-18
- At this point the only technique that uses the uveoscleral pathway is cyclodialysis. This surgical method separates the longitudinal ciliary muscle fibers from the adjacent scleral spur so a non-physiological pathway for aqueous humor drainage can be created. The cyclodialysis technique is generally combined with implants positioned ab-interno19,20
The common feature of all the surgical techniques mentioned above is the creation of an artificial channel to improve the outflow of aqueous humor and thus reduce intraocular pressure. This approach leads, firstly, to a compromise between the necessary drainage enhancement and the absence of complications. Secondly, there is an ongoing struggle against the natural healing processes to keep the artificially created drainage channel open as long as possible.
A new surgical method uses the uveoscleral pathway with an ab-externo access: this is the ‘cilio-scleral’ route. It does not create any artificial egress but locally alleviates the main obstacle to the aqueous drainage circulation in the supraciliary space to take advantage of the natural uveoscleral outflow potency.22
With this new method:
- The anterior chamber remains untouched, so there is:
- No risk of Endothelial Cell Loss (ECL)
- A marked reduction in the postoperative inflammation that is present in other glaucoma operations
- There is no bleb but still the IOP lowering effect is important
- Incisional trauma to the conjunctiva is limited and the anatomical structures are not damaged
- Another important aspect is that this technique is angle agnostic and can be used for Shaffer grades 1 to 421
References
- Bicket AK et al. (2021). Minimally Invasive Glaucoma Surgical Techniques for Open-Angle Glaucoma: An Overview of Cochrane Systematic Reviews and Network Meta-analysis. JAMA Ophthalmol. 2021 Sep 1;139(9):983-989
- Friedman DS et al. (2004). Eye Diseases Prevalence Research Group. Prevalence of open-angle glaucoma among adults in the United States. Arch Ophthalmol. 2004;122(4):532-538
- Quigley HA et al. (2006). The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006;90(3):262-267
- Tham YC et al. (2014). Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology. 2014;121(11):2081-2090
- Sun Y et al. (2022). Time trends, associations and prevalence of blindness and vision loss due to glaucoma: an analysis of observational data from the Global Burden of Disease Study 2017. BMJ Open. 2022 Jan 6;12(1):e053805
- Li T et al. (2016). Comparative effectiveness of first-line medications for primary open-angle glaucoma: a systematic review and network meta-analysis. Ophthalmology. 2016;123(1):129-140
- Bhartiya S. et al. (2015). Surgery on the Trabecular Meshwork: Histopathological Evidence. J Curr Glaucoma Pract. 2015 May-Aug;9(2):51-61
- Alm A. et al. (2009). Uveoscleral outflow—a review. Exp Eye Res. 2009;88(4):760
- Townsend DJ. et al. (1980). Immediate effect of epinephrine on aqueous formation in the normal human eye as measured by fluorophotometry. Invest Ophthalmol Vis Sci. 1980;19(3):256
- https://eyewiki.aao.org/Unconventional_Aqueous_Outflow
- Toris CB. et al. (1999). Aqueous humor dynamics in the aging human eye. Am J Ophthalmol. 1999 Apr;127(4):407-12
- Sunderland et al. (2023). Physiology, Aqueous Humor Circulation. StatPearls, 2023
- Skuta GL. et al. (1987). Wound healing in glaucoma filtering surgery. Surv Ophthalmol. 1987 Nov-Dec;32(3):149-70
- https://eyewiki.aao.org/Ab_Interno_Trabeculectomy_and_Trabeculotomy
- Bentley MD, Hann CR, Fautsch MP. Anatomical Variation of Human Collector Channel Orifices. Invest Ophthalmol Vis Sci. 2016 Mar;57(3):1153-9
- Shalaby WS. et al. (2021). iStent versus iStent inject implantation combined with phacoemulsification in open angle glaucoma. Indian J Ophthalmol. 2021 Sep;69(9):2488-2495
- Dickerson JE. et al. (2020). Circumferential canal surgery: a brief history. Curr Opin Ophthalmol. 2020 Mar;31(2):139-146
- Arriola-Villalobos P. et al. (2016). Glaukos iStent inject® Trabecular Micro-Bypass Implantation Associated with Cataract Surgery in Patients with Coexisting Cataract and Open-Angle Glaucoma or Ocular Hypertension: A Long-Term Study. J Ophthalmol. 2016;2016:1056573
- Pillunat LE. et al. (2017). Micro-invasive glaucoma surgery (MIGS): a review of surgical procedures using stents. Clin Ophthalmol. 2017 Aug 29;11:1583-1600
- González-Martín-Moro J. et al. (2017). Cyclodialysis: an update. Int Ophthalmol. 2017 Apr;37(2):441-457
- https://www.cilia.tech
- Emi K, Pederson JE, Toris CB. Hydrostatic pressure of the suprachoroidal space. Invest Ophthalmol Vis Sci. 1989 Feb;30(2):233-8