Scientific Resources
- News
First independent observations, presented by Dr. Leon Au, leading consultant ophthalmology and eye surgeon, during ICGS 2024 in Abu Dhabi, underscore CID’s strong potential as treatment option for glaucoma.
- News
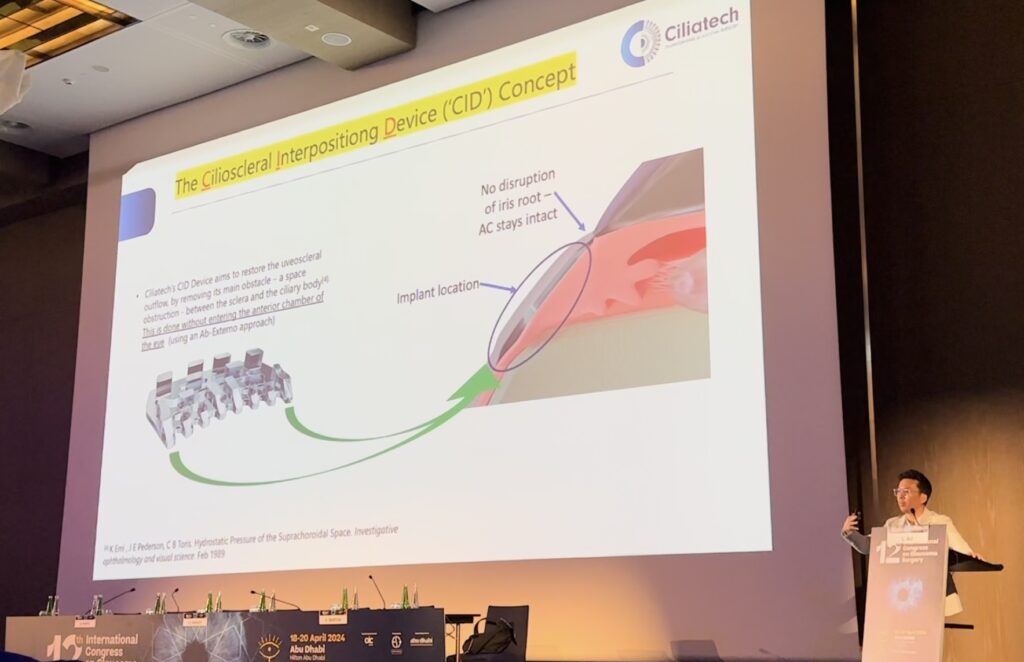
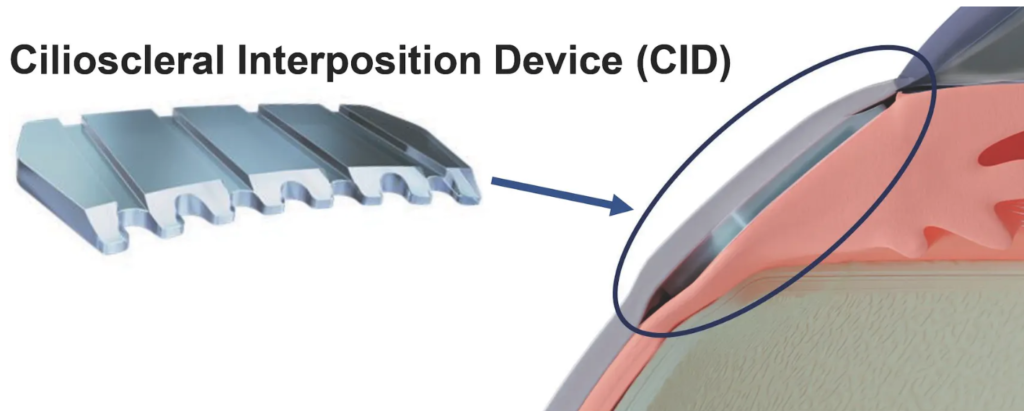
Second-generation CID (Cilioscleral Interpositioning Device) makes cilioscleral surgical procedure, which leaves anterior chamber of eye untouched, less invasive and easier to perform.
- Technical Articles
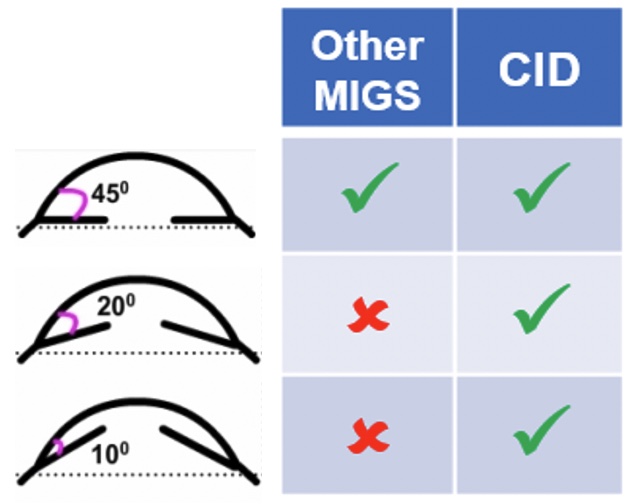
Primary angle-closure glaucoma (PACG) represents a significant cause of blindness, affecting more than 17 million people worldwide. Moreover, the currently available treatment options for the management PACG remain relatively limited. The cilioscleral inter-positioning device is a novel minimally invasive implant designed to increase uveoscleral outflow without entering the chamber. This unique approach to lower IOP represents a new, efficient, and safe surgical option to reduce intraocular pressure for patients with PACG.
- News
Ciliatech presents preliminary one-year study results on treating open and narrow angle glaucoma with CID at Ophthalmology Futures Symposium.
- News
At the World Glaucoma Congress (WGC) in Rome, Italy this summer, Ciliatech presented preliminary results of the second 2-year postoperative clinical trial (SAFARI Ⅱ) on its Cilioscleral Interposition Device (CID).
- Technical Articles
Glaucoma is a group of progressive, neurodegenerative eye diseases associated with irreversible optic neuropathy followed by visual-field impairment. Epidemiological estimates suggest that the most common form,
- News
Results from second CID (Cilio-scleral Interposition Device) study confirm sustained decrease in intraocular pressure (IOP), while preserving anterior chamber of eye
- News
You will be the 2nd employee of the Company, reporting directly to the CEO, and in close connection withe the medical Director...
- News